The Truth About Saltwater Pools
How Salt Chlorine Generators Really Work
Pool professionals hear the same statement constantly:
“I don’t use chlorine. I have a saltwater pool.”
This belief is one of the most persistent misconceptions in the swimming pool industry. The truth is straightforward: saltwater pools absolutely use chlorine. The only difference between a saltwater pool and a traditional chlorine pool is how the chlorine is delivered to the water.
Traditional pools rely on chlorine products such as liquid sodium hypochlorite, calcium hypochlorite, or trichlor tablets. Saltwater pools, on the other hand, produce chlorine inside the pool system itself using a device called a salt chlorine generator (SCG).
Understanding this chemistry is essential knowledge for professional pool operators and for anyone studying for Certified Pool Operator (CPO®) certification.
What Is a Saltwater Pool?
A saltwater pool is a swimming pool that uses dissolved salt and an electrolytic chlorine generator to produce chlorine sanitizer through electrochemical reactions.
The chlorine produced forms hypochlorous acid (HOCl), which is the same disinfectant responsible for killing bacteria, viruses, and algae in all properly sanitized swimming pools.
From a chemical standpoint, a saltwater pool is simply a chlorine pool with an automated chlorine production system.
What Is in a Saltwater Pool?
Saltwater pools contain sodium chloride (NaCl) dissolved in water. Sodium chloride is the same compound commonly known as table salt.
Chemically, sodium chloride consists of two elements:
- Sodium (Na)
- Chlorine (Cl)
Chlorine belongs to the halogen group on the periodic table, a family of elements known for their strong oxidizing and disinfecting properties.
When sodium chloride dissolves in water, it separates into ions:NaCl→Na++Cl−NaCl → Na^+ + Cl^-
This dissociation of ionic compounds in water is a fundamental concept in physical chemistry and is described in standard chemistry texts such as Physical Chemistry by Atkins and de Paula.
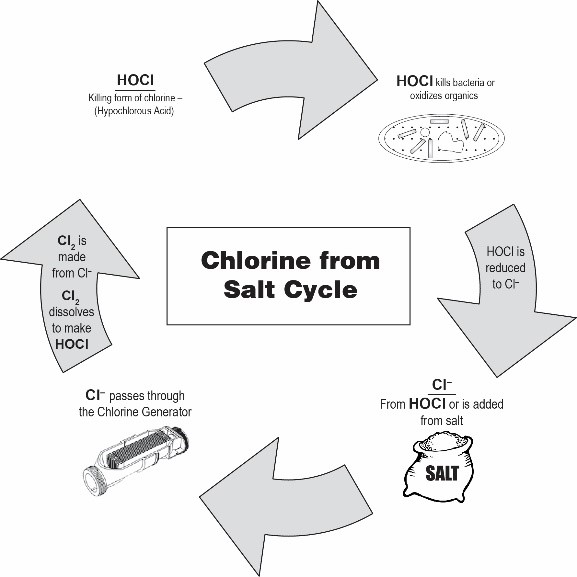
The chloride ion (Cl⁻) is the key component used by salt chlorine generators to produce chlorine.
How Salt Chlorine Generators Produce Chlorine
Inside a salt chlorine generator is an electrolytic cell containing electrically charged plates called the anode and cathode.
When saltwater flows through the cell and electricity is applied, a process called electrolysis occurs.
Electrolysis uses electrical energy to drive chemical reactions that would not otherwise occur spontaneously.
At the anode, chloride ions are oxidized to form chlorine gas:2Cl−→Cl2+2e−2Cl^- → Cl_2 + 2e^-
This electrochemical reaction is the same fundamental process used in industrial chlorine production within the chlor-alkali industry.
Once chlorine gas forms, it reacts immediately with water:Cl2+H2O⇌HOCl+H++Cl−Cl_2 + H_2O ⇌ HOCl + H^+ + Cl^-
This reaction produces hypochlorous acid (HOCl).
Hypochlorous acid and its conjugate base, hypochlorite ion (OCl⁻), together form what pool operators measure as Free Available Chlorine (FAC).
The disinfecting chemistry of chlorine in water is extensively documented in water treatment research, including Handbook of Chlorination and Alternative Disinfectants by George Clifford White.
Electrolysis of Water Inside the Salt Cell
Water itself also participates in electrolysis reactions inside the chlorine generator.
At the cathode, water molecules are reduced to form hydrogen gas and hydroxide ions:2H2O+2e−→H2+2OH−2H_2O + 2e^- → H_2 + 2OH^-
Hydrogen gas escapes from the cell as bubbles that can often be seen inside operating salt chlorine generators.
These reactions are well studied in electrochemical engineering and are described in Electrochemical Methods: Fundamentals and Applications by Bard and Faulkner.
Why Saltwater Pools Often Experience Rising pH
One of the common operational characteristics of saltwater pools is gradual pH rise over time.
This occurs because electrolysis produces hydroxide ions (OH⁻) at the cathode. Hydroxide ions increase alkalinity locally and contribute to upward pH drift.
In addition, hydrogen gas produced during electrolysis escapes from the water. When hydrogen gas leaves the system, it removes acidity and contributes to further pH increase.
Studies examining electrolytic chlorination systems confirm that pH rise is a typical operational effect of salt chlorine generators.
Because of this, saltwater pools frequently require periodic additions of muriatic acid to maintain proper water balance.
Saltwater Pool vs Chlorine Pool
| Feature | Saltwater Pool | Traditional Chlorine Pool |
|---|---|---|
| Primary Sanitizer | Chlorine | Chlorine |
| Chlorine Delivery | Produced through electrolysis of salt | Added manually |
| Sanitizing Chemical | Hypochlorous acid (HOCl) | Hypochlorous acid (HOCl) |
| Salt Level | ~3,000–4,000 ppm | Typically <500 ppm |
| Maintenance | Requires water testing and balancing | Requires water testing and balancing |
Both systems rely on the same disinfecting chemistry. The difference is simply whether chlorine is generated automatically or added manually.
Salt Levels in a Saltwater Pool
Most residential salt chlorine generators operate at salt concentrations between 3,000 and 4,000 ppm.
In a 10,000-gallon pool, this requires approximately 250 to 330 pounds of salt.
For comparison:
| Water Type | Average Salt Concentration |
|---|---|
| Saltwater Pool | 3,000–4,000 ppm |
| Human Tears | ~7,000 ppm |
| Ocean Water | ~35,000 ppm |
This means saltwater pools contain less than one-tenth the salinity of seawater, which is why the water tastes only mildly salty.
Saltwater Pools and Total Dissolved Solids
Adding salt increases the Total Dissolved Solids (TDS) level in the water.
In pool chemistry calculations, a useful rule is:
1 pound of dissolved material added to 10,000 gallons increases TDS by approximately 12 ppm.
Therefore, adding 300 pounds of salt increases TDS by roughly 3600 ppm.
This relationship comes from basic concentration calculations used in environmental water chemistry and wastewater engineering.
Similar Article Total Dissolved Solids in Swimming Pools
Does Salt Affect pH?
Another common misconception is that salt itself raises pH.
In reality, pure sodium chloride solutions are nearly neutral, with a pH close to 7.
This occurs because sodium chloride forms from a strong acid (hydrochloric acid) and a strong base (sodium hydroxide). Salts formed from strong acids and strong bases generally produce neutral aqueous solutions.
The rising pH observed in saltwater pools therefore comes from electrochemical reactions in the chlorine generator, not from the salt itself.
Why Some People Think Saltwater Pools Are Chlorine Free
When salt chlorine generators first became popular, they were often marketed as an alternative to chlorine pools.
Some marketing materials suggested that saltwater pools would eliminate chlorine or create chemical-free water.
These claims were misleading.
Saltwater systems do reduce the need to manually add chlorine products, but the sanitizer produced in the water is still chlorine in the form of hypochlorous acid.
From a chemistry perspective, a saltwater pool is simply a chlorine pool that produces its chlorine automatically.
CPO Study Guide: Saltwater Pool Chemistry
For students preparing for Certified Pool Operator (CPO®) certification, several key principles should be remembered about saltwater pools.
Saltwater pools sanitize water using chlorine chemistry, just like traditional chlorine pools.
Salt chlorine generators produce chlorine through electrolysis of dissolved sodium chloride.
The chlorine produced forms hypochlorous acid (HOCl), the same disinfectant responsible for pathogen control in all chlorinated swimming pools.
Electrolysis also produces hydroxide ions and hydrogen gas, which is why saltwater pools commonly experience gradual pH rise.
Understanding these reactions is essential for maintaining balanced, safe swimming pool water.
Frequently Asked Questions About Saltwater Pools
Are saltwater pools chlorine free?
No. Saltwater pools produce chlorine through electrolysis. The sanitizer produced is hypochlorous acid, the same disinfectant used in traditional chlorine pools.
Is a saltwater pool safer than a chlorine pool?
Both systems rely on chlorine chemistry to disinfect the water. The sanitizer responsible for killing pathogens is the same compound: hypochlorous acid.
Do saltwater pools require less maintenance?
Salt systems automate chlorine production, but pools still require regular water testing, balancing, and cleaning.
Why does pH rise in saltwater pools?
Electrolysis produces hydroxide ions and hydrogen gas, both of which contribute to gradual pH increase.
Do saltwater pools taste like ocean water?
No. Ocean water averages about 35,000 ppm salt, while saltwater pools operate around 3,000–4,000 ppm, making the water only mildly salty.
Learn More About Pool Water Chemistry
Understanding saltwater pool chemistry is only one part of maintaining safe aquatic environments.
Professional pool operators and facility managers responsible for water safety can learn more about water balance, sanitation chemistry, filtration, and risk management through Certified Pool Operator (CPO®) certification training.
Virtual CPO classes taught by Rudy Stankowitz provide in-depth instruction designed specifically for pool professionals responsible for safe and compliant pool operation.
Academic and Technical References
Atkins, P., & de Paula, J. Physical Chemistry. Oxford University Press.
Bard, A., & Faulkner, L. Electrochemical Methods: Fundamentals and Applications. Wiley.
Brown, T., LeMay, H., Bursten, B. Chemistry: The Central Science. Pearson.
Chen, X. et al. Electrochemical reactions in salt chlorine generators. Electrochimica Acta.
White, G. Handbook of Chlorination and Alternative Disinfectants. Wiley.
World Health Organization. Guidelines for Safe Recreational Water Environments.